Chemical and Physical Reactions Test
Page 1
1. Which are all chemical properties (no physical properties)?
a. color, size, shape, and mass
b. flammability, mass, and freezing point
c. size, pH, and boiling point
d. flammability, pH, reactivity
2. When a burning splint was put in the tube of gas collected above electrode B, the gas burned and produced a squeaky popping sound. Which gas has this chemical property of being flammable?
a. Air
b. Steam
c. Hydrogen
d. Oxygen
a. color, size, shape, and mass
b. flammability, mass, and freezing point
c. size, pH, and boiling point
d. flammability, pH, reactivity
2. When a burning splint was put in the tube of gas collected above electrode B, the gas burned and produced a squeaky popping sound. Which gas has this chemical property of being flammable?
a. Air
b. Steam
c. Hydrogen
d. Oxygen
3. Michael noticed that the iron metal on his new bike was starting to rust. He learned in science class that the scientific name for rust is iron oxide (Fe2O3). He also knew that:
a. Rust is an element that forms any time iron and water (H2O) mix
b. Rust is a compound that forms when oxygen combines with iron.
c. Iron combines with water and oxygen to make a mixture called rust.
d. Oxygen was released from the iron on his bike to form rust atoms.
4. Which is a clue that would describe a chemical change?
a. Breaking
b. Boiling
c. Freezing
d. Burning
5. What characteristic best describes what happens during a physical change?
a. The elements in the substance change.
b. The elements in the substance stay the same.
c. The elements in the substance re-arrange.
d. The elements in the substance decompose.
6. When a new substance is formed with different properties than the original substance it is called…
a. a chemical change
b. a physical change
c. freezing
d. boiling
a. Rust is an element that forms any time iron and water (H2O) mix
b. Rust is a compound that forms when oxygen combines with iron.
c. Iron combines with water and oxygen to make a mixture called rust.
d. Oxygen was released from the iron on his bike to form rust atoms.
4. Which is a clue that would describe a chemical change?
a. Breaking
b. Boiling
c. Freezing
d. Burning
5. What characteristic best describes what happens during a physical change?
a. The elements in the substance change.
b. The elements in the substance stay the same.
c. The elements in the substance re-arrange.
d. The elements in the substance decompose.
6. When a new substance is formed with different properties than the original substance it is called…
a. a chemical change
b. a physical change
c. freezing
d. boiling
Page 2
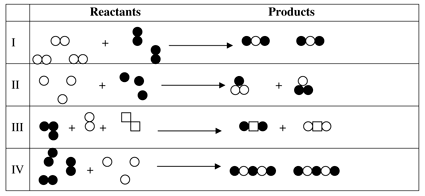
7. Reactants are the original substances that are used prior to a chemical reaction happening. Products are made by rearranging the reactants. In the process of a chemical reaction no amount of matter is lost it is only rearranged differently than it was before the reaction took place. By looking at the diagram below choose any combination of the options that shows a true chemical reaction took place.
a. II b. I, II, III c. I, III, IV d. I, II, III, IV
8. All elements contain a certain amount of energy. However when reactant elements are combined, and begin to rearrange, they begin to release energy. When a chemical reaction is taking place more and more energy is being released until the majority of elements are rearranged. By looking at the graph below:
1) Describe what happens to the amount of energy when the reactants are combined..
2) Compare the amount of energy reactants have versus the energy products have.
1) Describe what happens to the amount of energy when the reactants are combined..
2) Compare the amount of energy reactants have versus the energy products have.
Page 3
Reading passage
Several factors affect the rate (how fast the chemical reacts) at which a chemical reaction occurs. One factor that affects the rate of reaction is temperature.
When Solutions A and B (two colorless liquids) are mixed, a reaction takes place. When the reaction is completed, the mixture turns dark blue.
Experiment 1
Students mixed 20 ml each of Solutions A and B at 3 different temperatures. Each time, they stirred until the reaction was complete. The average reaction times for 5 trials are shown in the table.
Several factors affect the rate (how fast the chemical reacts) at which a chemical reaction occurs. One factor that affects the rate of reaction is temperature.
When Solutions A and B (two colorless liquids) are mixed, a reaction takes place. When the reaction is completed, the mixture turns dark blue.
Experiment 1
Students mixed 20 ml each of Solutions A and B at 3 different temperatures. Each time, they stirred until the reaction was complete. The average reaction times for 5 trials are shown in the table.
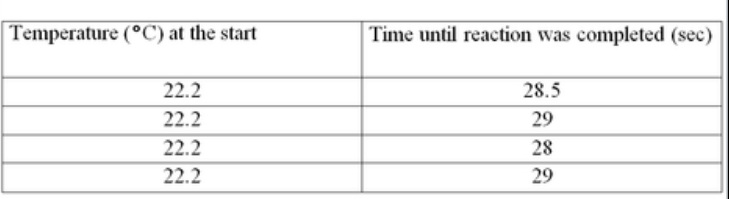
Experiment 2
The students mixed 20 ml each of Solutions A and B at 22.2 °C and stirred the mixture as the reaction proceeded. The students recorded the time that it took for the mixture to turn dark blue. This was repeated 4 more times.
The students mixed 20 ml each of Solutions A and B at 22.2 °C and stirred the mixture as the reaction proceeded. The students recorded the time that it took for the mixture to turn dark blue. This was repeated 4 more times.
1. How is the experimental design of Experiment 1 different from that of Experiment 2?
a. Experiment 1 varies the temperature of the mixture and Experiment 2 has only one temperature.
b. Experiment 2 varies the temperature of the mixture and Experiment 1 has only one temperature.
c. Experiment 1 varies the time of the mixture and experiment 2 varies the temperature.
d. Experiment 1 and Experiment 2 have the same experimental design.
2. Which of the following conditions was directly changed by the students in Experiment 1?
a. Total volume of the mixture
b. Concentration of each solution in the mixture
c. Temperature of the mixture
d. Reaction rate
a. Experiment 1 varies the temperature of the mixture and Experiment 2 has only one temperature.
b. Experiment 2 varies the temperature of the mixture and Experiment 1 has only one temperature.
c. Experiment 1 varies the time of the mixture and experiment 2 varies the temperature.
d. Experiment 1 and Experiment 2 have the same experimental design.
2. Which of the following conditions was directly changed by the students in Experiment 1?
a. Total volume of the mixture
b. Concentration of each solution in the mixture
c. Temperature of the mixture
d. Reaction rate